EPIDEMIOLOGY
The geographic range of CCHF virus is the most extensive among the
tickborne viruses that affect human health, and the second most
widespread of all medically important arboviruses, after dengue viruses (Ergonul, 2006).
The disease was first discovered in the Crimean region of Russia in
the 1940s and is now reported in many regions of the world: Africa, the
Middle East, Europe and Asia. In the territory of the former Soviet
Union, disease outbreaks or the presence of the virus were reported in
the southern regions of European Russia, in Mouldova, Ukraine and
Transcaucasus, and in Central Asian countries, in Tajikistan,
Turkmenistan, Uzbekistan, Kyrgyzstan and Kazakhstan (Yashina et al., 2003).
Published descriptions of major epidemics and outbreaks of CCHF have
been reviewed extensively in the past. These reports illustrate the very
wide distribution of CCHF virus. This distribution stretches over much
of Asia, extending from the XinJiang region of China to the Middle East
and southern Russia, and to focal endemic areas over much of Africa and
parts of south-eastern Europe. Thus, CCHF virus is the most widely
distributed agent of severe haemorrhagic fever known (Chamberlain et al., 2005).
Before 1970, most cases were reported from the former Soviet Union
(Crimea, Astrakhan, Rostov, Uzbekistan, Kazakhstan, Tajikistan) and
Bulgaria, as well as virus circulation in parts of Africa such as the
Democratic Republic of the Congo and Uganda.
The initial recognition of haemorrhagic cases in Africa occurred in the 1960s, resulting in a series of in-depth studies in South Africa and reports of additional outbreaks from Congo, Mauritania, Burkina Faso, Tanzania, and Senegal. A substantial number of cases were also reported from middle eastern countries such as Iraq, the United Arab Emirates (UAE), Saudi Arabia and Oman, and from Pakistan and China. By 2000, new outbreaks had been reported from Pakistan, Iran, Senegal, Albania, Yugoslavia, Bulgaria, Turkey, Kenya, and Mauritania. Serological evidence for CCHF virus has been reported from Greece, India, Egypt, Portugal, Hungary, France, and Benin, although the virus was isolated only in Greece and the only reported human case was a Greek laboratory infection. CCHF virus is endemic in the Balkans, including Bulgaria, the former Yugoslavia, and Albania. It is of interest that the strain that caused the laboratory-related infection in Greece was exceedingly mild, possibly reflecting chance variation; however, the virus has the greatest phylogenetic difference from other CCHF viruses and Greece is separated from Bulgaria by mountains approximately 1500-2500 m high (Ergonul, 2006). The common vector for CCHF virus are ticks of the genus Hyalomma.
The virus is transmitted to humans either directly by Hyalomma ticks or by contact with infected domestic animals. CCHF virus is primarily a zoonosis, which means that the transmission cycle mainly involves ticks and wild or domestic animals. Cattle, sheep and goats do not become ill after infection but are viremic for about 1 week. During this period of time the virus may be transmitted to humans which have close contact to these animals such as agricultural workers, slaughterhouse workers, and veterinarians. Furthermore, the virus may be spread into other geographical regions via infected livestock. The virus may also be transmitted from human to human which occurs primarily in the hospital setting. Health care workers are mainly at risk (Drosten et al., 2003).
The initial recognition of haemorrhagic cases in Africa occurred in the 1960s, resulting in a series of in-depth studies in South Africa and reports of additional outbreaks from Congo, Mauritania, Burkina Faso, Tanzania, and Senegal. A substantial number of cases were also reported from middle eastern countries such as Iraq, the United Arab Emirates (UAE), Saudi Arabia and Oman, and from Pakistan and China. By 2000, new outbreaks had been reported from Pakistan, Iran, Senegal, Albania, Yugoslavia, Bulgaria, Turkey, Kenya, and Mauritania. Serological evidence for CCHF virus has been reported from Greece, India, Egypt, Portugal, Hungary, France, and Benin, although the virus was isolated only in Greece and the only reported human case was a Greek laboratory infection. CCHF virus is endemic in the Balkans, including Bulgaria, the former Yugoslavia, and Albania. It is of interest that the strain that caused the laboratory-related infection in Greece was exceedingly mild, possibly reflecting chance variation; however, the virus has the greatest phylogenetic difference from other CCHF viruses and Greece is separated from Bulgaria by mountains approximately 1500-2500 m high (Ergonul, 2006). The common vector for CCHF virus are ticks of the genus Hyalomma.
The virus is transmitted to humans either directly by Hyalomma ticks or by contact with infected domestic animals. CCHF virus is primarily a zoonosis, which means that the transmission cycle mainly involves ticks and wild or domestic animals. Cattle, sheep and goats do not become ill after infection but are viremic for about 1 week. During this period of time the virus may be transmitted to humans which have close contact to these animals such as agricultural workers, slaughterhouse workers, and veterinarians. Furthermore, the virus may be spread into other geographical regions via infected livestock. The virus may also be transmitted from human to human which occurs primarily in the hospital setting. Health care workers are mainly at risk (Drosten et al., 2003).
Environmental Reservoir:

RESERVOIRS FOR CCHF
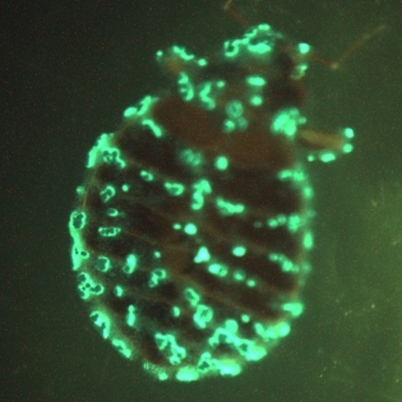
Ticks:-
Description: While only a few species of ticks have been incriminated as vectors of CCHF virus, an enormous number of species/subspecies have been implicated primarily by viral isolations. In 1973, only 6 years after the first isolations of CCHF virus, a total of 10 species/subspecies had yielded isolates of this virus. A remarkable and especially important epidemiological feature that emerged was not only the large number of implicated vector tick species, but the association of CCHF virus with ticks in a variety of different ecological biotypes in the Palearctic, Oriental, and Ethiopian faunal regions. Among the total 29 species/subspecies of ticks associated with CCHF virus, most are either two- or three-host ticks of the family Ixodidae. An especially important biological feature of ticks in general as potential vector/reservoirs of arboviruses is their ability to transmit arboviruses transovarially. Evidence of this phenomenon for CCHF virus in nature is based mainly on limited isolations from eggs of H. marginatum and Dermacentor marginatus (Watts et al., 1988). Most of the ticks collected (618 of 912) from all species of sampled livestock were Hyalomma anatolicum anatolicum, a competent vector and reservoir of CCHF virus (Williams et al., 2000).
The fever spreads to humans either by tick-bites, or through contact with viremic animal tissues during or immediately after slaughter. CCHF outbreaks constitute a threat to public health services because of its epidemic potential and its high case fatality ratio
Description: While only a few species of ticks have been incriminated as vectors of CCHF virus, an enormous number of species/subspecies have been implicated primarily by viral isolations. In 1973, only 6 years after the first isolations of CCHF virus, a total of 10 species/subspecies had yielded isolates of this virus. A remarkable and especially important epidemiological feature that emerged was not only the large number of implicated vector tick species, but the association of CCHF virus with ticks in a variety of different ecological biotypes in the Palearctic, Oriental, and Ethiopian faunal regions. Among the total 29 species/subspecies of ticks associated with CCHF virus, most are either two- or three-host ticks of the family Ixodidae. An especially important biological feature of ticks in general as potential vector/reservoirs of arboviruses is their ability to transmit arboviruses transovarially. Evidence of this phenomenon for CCHF virus in nature is based mainly on limited isolations from eggs of H. marginatum and Dermacentor marginatus (Watts et al., 1988). Most of the ticks collected (618 of 912) from all species of sampled livestock were Hyalomma anatolicum anatolicum, a competent vector and reservoir of CCHF virus (Williams et al., 2000).
The fever spreads to humans either by tick-bites, or through contact with viremic animal tissues during or immediately after slaughter. CCHF outbreaks constitute a threat to public health services because of its epidemic potential and its high case fatality ratio
PATHOPHYSIOLOGY
The main common underlying pathophysiologic feature of viral
hemorrhagic fevers is that the vascular bed is attacked, with resultant
microvascular damage and changes in vascular permeability. However,
specific pathophysiologic findings can vary depending on the virus
family and the species involved.
In general, an initial febrile illness is followed by hemorrhaging into the skin and the mucous membranes; hemorrhagic rashes; and hemorrhages from body orifices, especially gastrointestinal and genitourinary bleeding. Lassa fever, although fatal, is not characterized by significant bleeding. Other clinical findings include thrombocytopenia and leukocytopenia
In general, an initial febrile illness is followed by hemorrhaging into the skin and the mucous membranes; hemorrhagic rashes; and hemorrhages from body orifices, especially gastrointestinal and genitourinary bleeding. Lassa fever, although fatal, is not characterized by significant bleeding. Other clinical findings include thrombocytopenia and leukocytopenia
TRANSMISSION
CCHFV usually circulates between asymptomatic animals and ticks in an enzootic cycle. This virus has been found in at least 31 species of ticks, including seven genera of the family Ixodidae (hard ticks). Members of the genus Hyalomma seem to be the principal vectors. Transovarial, transstadial and venereal transmission occur in this genus. Hyalomma marginatum marginatum is particularly important as a vector in Europe, but CCHFV is also found in Hyalomma anatolicum anatolicum and other Hyalomma spp. Other ixodid ticks including members of the genera Rhipicephalus, Boophilus, Dermacentor and Ixodes may also transmit the virus locally. Although CCHFV has been reported in other families of invertebrates, these species may not be biological vectors; the virus may have been ingested in a recent blood meal. In one study, CCHFV was reported from a biting midge (Culicoides spp.). It has also been found in two species of Argasidae (soft ticks); however, experimental infections suggest that CCHFV does not replicate in this family of ticks.
Many species of mammals can transmit CCHFV to ticks when they are viremic. Small vertebrates such as hares and hedgehogs, which are infested by immature ticks, may be particularly important as amplifying hosts. With a few exceptions, birds seem to be refractory to infection; however, they may act as mechanical vectors by transporting infected ticks. Migratory birds might spread the virus between distant geographic areas.
Humans become infected through the skin and by ingestion. Aerosol transmission was suspected in a few cases in Russia. Sources of exposure include being bitten by a tick, crushing an infected tick with bare skin, contacting animal blood or tissues and drinking unpasteurized milk. Human-to-human transmission occurs, particularly when skin or mucous membranes are exposed to blood during hemorrhages or tissues during surgery. CCHFV is stable for up to 10 days in blood kept at 40°C (104°F). Possible horizontal transmission has been reported from a mother to her child.
INCUBATION PERIOD
The incubation period is influenced by the route of exposure. Infections acquired via tick bites usually become apparent after 1 to 3 days; the longest incubation period reported by this route is nine days. Exposure to blood or tissues usually results in a longer incubation period. Current estimates suggest that these infections become apparent, on average, after 5 to 6 days, but incubation periods up to 13 days are known
SYMPTOMS
Clinical Signs
The first sign of Crimean-Congo hemorrhagic fever is a sudden onset of fever and other nonspecific symptoms including chills, severe headache, dizziness, photophobia, neck pain, myalgia and arthralgia. The fever may be very high. Gastrointestinal symptoms including nausea, vomiting, non-bloody diarrhea and abdominal pain are also common. Sharp mood changes, confusion and aggression have been reported in some cases. Cardiovascular changes such as bradycardia and low blood pressure can also occur. This early stage of disease is called the prehemorrhagic phase. It is followed, after several days, by the hemorrhagic phase.
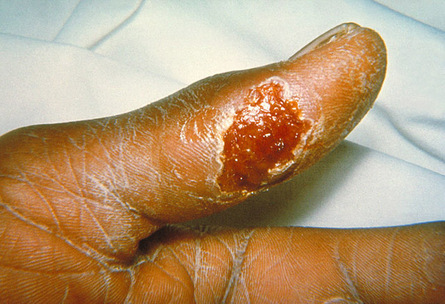
The hemorrhagic phase develops suddenly. It is usually short, lasting on average 2 to 3 days. A petechial rash may be the first symptom. The rash is followed by petechiae, ecchymoses and large bruises on the skin and mucous membranes. Hematemesis, melena, epistaxis, hematuria, hemoptysis and bleeding from venipuncture sites are also common. Bleeding can occur in other locations, including the brain. In one case, internal bleeding mimicked acute appendicitis. Hepatitis occurs in some patients, and may result in jaundice and hepatomegaly. Splenomegaly can also be seen. Some patients die from hemorrhages, hemorrhagic pneumonia or cardiovascular disturbances.
In patients who survive, recovery begins 10 to20 days after the onset of illness. The convalescent phase is characterized by generalized weakness, a weak pulse and tachycardia. Other symptoms including sweating, dryness of the mouth, headache, dizziness, nausea, poor appetite, labored breathing, polyneuritis, poor vision, loss of hearing, and memory loss have also be seen. Some patients temporarily lose all of their hair. Hepatorenal insufficiency has been reported in some countries but not others. Recovery is usually complete but slow, and can take up to a year. Subclinical infections can occur, but are thought to be uncommon. Mild febrile cases without hemorrhages are also seen.
DISINFECTION
CCHFV can be inactivated by disinfectants including 1% hypochlorite and 2% glutaraldehyde. It is also destroyed by heating at 56°C (133°F) for 30 min.
DIAGNOSIS
Although the clinical findings can suggest a hemorrhagic fever,
laboratory studies are required to identify disease, to distinguish it
from other conditions, and to confirm its etiology.
As a rule, clinical blood and/or urine tests reveal leukopenia (except in Lassa fever, Hantaan viral fever, and some severe cases of CCHF), thrombocytopenia (except in Lassa fever), and proteinuria and/or hematuria (in Argentinian viral hemorrhagic fever, Bolivian viral hemorrhagic fever, and hemorrhagic fever with renal syndrome [HFRS]; common in other viral hemorrhagic fevers). Such tests include the following:
Specialized infectious disease containment is required for the safe handling of these viruses. Biochemical tests are available for the rapid detection of viral antigen during viremia or in postmortem specimens. Such tests include the following:
As a rule, clinical blood and/or urine tests reveal leukopenia (except in Lassa fever, Hantaan viral fever, and some severe cases of CCHF), thrombocytopenia (except in Lassa fever), and proteinuria and/or hematuria (in Argentinian viral hemorrhagic fever, Bolivian viral hemorrhagic fever, and hemorrhagic fever with renal syndrome [HFRS]; common in other viral hemorrhagic fevers). Such tests include the following:
- Full blood cell count determination
- Red cell and sedimentation rate analysis
- White cell differential count determination
- Platelet count
- Tests for parasites, malaria, trypanosomiasis, or relapsing fever
- Coagulation studies
- Liver and kidney function tests
- Bacterial culture and virus isolation from feces and urine samples
- Electron microscopic examination of blood and urine samples
- Positive tourniquet test in dengue fever (This test may also be helpful in other viral hemorrhagic fevers).
- The specific diagnosis is based on viral isolation or the demonstration of seroconversion, that is, the presence of specific immunoglobulin M (IgM) antibody or a 4-fold increase in the antibody titer.
Specialized infectious disease containment is required for the safe handling of these viruses. Biochemical tests are available for the rapid detection of viral antigen during viremia or in postmortem specimens. Such tests include the following:
- Immunofluorescence antibody (IFA) tests can be performed.
- Reversed (or indirect) passive hemagglutination (RPHA) tests may be helpful.
- Acute serum samples can be tested by using a polymerase chain reaction (PCR).
- Enzyme-linked immunosorbent assays (ELISA) can be used to detect specific IgM or immunoglobulin G (IgG) antibodies or viral antigens in acute serum samples from patients with Lassa fever, Argentinian fever, RVF, CCHF, or yellow fever. Lassa- and Hantavirus-specific IgM antibodies are often detectable during acute illness.
- Tests for viral genetic material are favored in diagnoses of acute arenaviral infections.
- Virus cultivation and identification techniques require 3-10 days or longer for definitive identification. Viral isolation is a lengthy process, especially in Hantaan, Ebola, Marburg, and Congo-Crimean fevers.
- Postmortem immunocytochemical analyses can be used to identify the viral phenotype.
- Reverse transcriptase loop-mediated isothermal amplification (RT-LAMP) can be used for the rapid detection of Marburg virus.11
TREATMENT
Treatment is mainly supportive. Ribavirin ( Rebetol) is used in some cases. Observational studies in humans and studies in experimentally infected mice support the use of this drug; however, no randomized human clinical trials have been published. Passive immunotherapy with hyperimmune serum has been tested in a few cases, but the value of this treatment is controversial.
TO KNOW MORE ON...........
RIBAVIRIN REBETOL http://www.rxlist.com/rebetol-drug.htm
RIBAVIRIN REBETOL http://www.rxlist.com/rebetol-drug.htm